Are the serum delta neutrophil index and systemic inflammatory index useful as predictive parameters for preeclampsia and HELLP syndrome?
Jsou sérový neutrofilní delta index a systémový zánětlivý index užitečné jako prediktivní parametry preeklampsie a HELLP syndromu?
Cíl: Naším cílem bylo zjistit, zda sérový delta index neutrofilů a další parametry systémového zánětlivého indexu mohou mít při použití s jinými bio chemickými markery u preeklampsie a HELLP syndromu pomocný účinek v diagnostice a určit roli zánětu v patogenezi těchto onemocnění. Materiál a metody: Do studie bylo zahrnuto 121 těhotných žen, které splnily kritéria pro zařazení a vyloučení. Do sledované skupiny bylo zařazeno 52 těhotných s diagnostikovanou preeklampsií, 19 těhotných s diagnostikovaným HELLP syndromem a do kontrolní skupiny 50 zdravých těhotných žen. Demografické údaje, hematologické a bio chemické parametry a zánětlivé markery (sérový neutrofilní delta index – DNI – a systémové parametry zánětlivého indexu) skupin byly zaznamenány a porovnány mezi skupinami. Výsledky: Z hlediska poměru neutrofilních lymfocytů, poměru lymfocytů trombocytů a DNI byla skupina HELLP odlišná od obou skupin. Kontrolní skupina a skupina s preeklampsií byly podobné. Z hlediska poměru monocytů k lymfocytům byla skupina preeklampsie odlišná od obou skupin. Kontrolní a HELLP skupiny byly podobné. Z hlediska indexu systémového zánětu byly všechny skupiny podobné. Závěr: V naší studii jsme zjistili, že využití DNI v mateřském séru spolu s dalšími bio chemickými parametry může pomoci v diagnostice preeklampsie a HELLP syndromu a zánět může hrát roli v patogenezi těchto onemocnění.
Authors:
Yusuf Dal 1
; Şebnem Karagün 1
; Fatih Akkuş 2
; K. Akay 3
; Ayhan Coşkun 1
Authors‘ workplace:
Department of Obstetrics and Gynecology, Perinatology Department, Mersin University Faculty of Medicine, Mersin, Turkey
1; Department of Obstetrics and Gynecology, Perinatology Department, Necmettin Erbakan University Meram Faculty of Medicine, Konya, Turkey
2; Department of Obstetrics and Gynecology, Mersin University Faculty of Medicine, Mersin, Turkey
3
Published in:
Ceska Gynekol 2023; 88(6): 412-419
Category:
Original Article
doi:
https://doi.org/10.48095/cccg2023412
Overview
Objective: We aimed to determine whether the serum delta neutrophil index and other systemic inflammatory index parameters can have an auxiliary effect in the diagnosis when used with other bio chemical markers in preeclampsia and HELLP syndrome and to determine the role of inflammation in the pathogenesis of these diseases. Materials and methods: 121 pregnant women who met the inclusion and exclusion criteria were included in the study. 52 pregnant women diagnosed with preeclampsia and 19 pregnant women diagnosed with HELLP syndrome were included in the study group, and 50 healthy pregnant women were included in the control group. Demographic data, hematological and bio chemical parameters, and inflammatory markers (serum delta neutrophil index – DNI – and systemic inflammatory index parameters) of the groups were recorded and compared between groups. Results: In terms of neutrophil lymphocyte ratio, platelet lymphocyte ratio, and DNI, the HELLP group was different from both groups. The control and preeclampsia groups were similar. In terms of monocyte-to-lymphocyte ratio, the preeclampsia group was different from both groups. The control and HELLP groups were similar. In terms of the systemic inflammatory index, all groups were similar. Conclusion: In our study, we found that when maternal serum DNI values are used together with other bio chemical parameters, it can help in the diagnosis of preeclampsia and HELLP syndrome, and inflammation may play a role in the pathogenesis of these diseases.
Keywords:
HELLP syndrome – preeclampsia – systemic inflammatory index – delta neutrophil index
Introduction
Hypertensive disorders of pregnancy (HDP) are an important cause of maternal and perinatal morbidity and mortality. HDP can be seen in approximately 10% of all pregnancies and can occur at any time of pregnancy [1].
Many factors are considered in the pathogenesis of HDP. The exact etiological factor is still unclear. Nulliparity, maternal age, insulin resistance, subclinical infections, a calcium-poor diet, metabolic syndrome, genetic predisposition, or immune factors alone or together increase the risk of HDP [2].
The pathogenesis of preeclampsia and HELLP syndrome is not fully understood. It is thought that excessive systemic inflammatory responses may play a role in the pathogenesis [3]. The products resulting from an increased inflammatory response may cause abnormal placentation, syncytiotrophoblast ischemia, and endothelial dysfunction [4]. These activated inflammatory cells can invade vascular areas with endothelial damage. As a result, it may contribute to increased endothelial dysfunction and thrombotic and metabolic disorders [5].
The serum delta neutrophil index (DNI), a new inflammatory marker, reflects the ratio of circulating immature granulocytes to the total neutrophil count in cases of infection and inflammation [6]. Systemic inflammatory cell indices are derived from peripheral blood cells and can be easily measured. For this reason, many studies have been carried out on this subject in recent years. Many obstetric conditions such as gestational diabetes, preterm premature rupture of membranes (PPROM), preterm birth, hyperemesis gravidarum, cesarean scar pregnancies, and intrahepatic cholestasis of pregnancy have been associated with inflammation [7–11]. The role of these systemic inflammatory indices (SII) in the diagnosis and prognosis assessment of HDP is not clear.
Preeclampsia and HELLP syndrome are progressive and unpredictable. Timely termination of pregnancy is the only treatment, so early diagnosis and follow-up are important. Although endothelial dysfunction and increased vascular resistance have been proven in preeclampsia, a direct relationship with inflammation has not been proven. DNI and other SII parameters, which are new inflammatory markers, are low-cost, easily accessible, and easily calculated with routine blood tests. There are few studies in the literature about the effects of these parameters on the diagnosis and prognosis of preeclampsia. However, there are no studies on the more serious form of the disease, HELLP syndrome (hemolysis, elevated liver enzymes, and low platelet count syndrome). The challenge of managing these diseases remains, and more research is also needed. A clear understanding of the pathogenesis and targeted therapy is important for the follow-up and treatment of these diseases. Our aim in this study was to evaluate the systemic inflammatory response in preeclampsia and HELLP syndrome and to determine the effect of SII and a new marker, DNI, in the diagnosis of these diseases.

*One Way ANOVA Test, **Kruskal Wallis Test, ***Post-hoc Bonferroni Test, ****Bonferroni-corrected Mann-Whitney U-Test.
Bold P-values indicate statistically signifi cant.
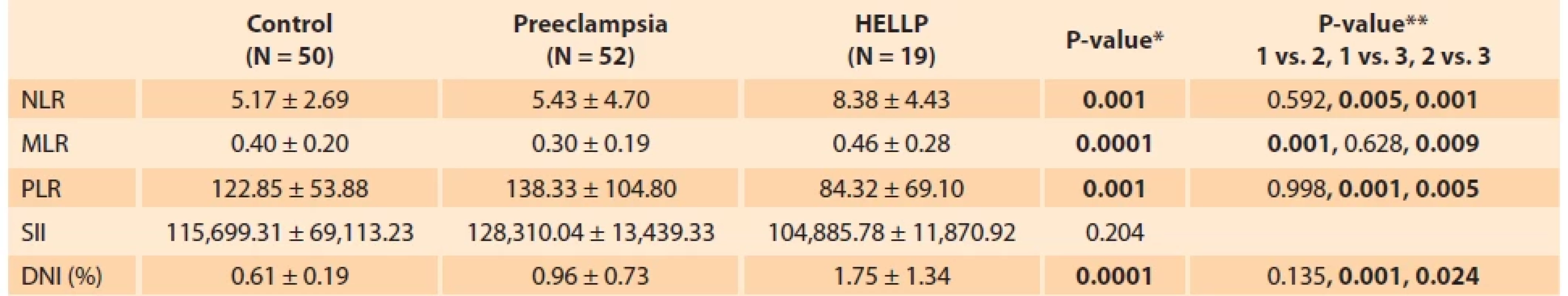
ratio, SII – systemic infl ammatory indices
SD – standard deviation is signifi cant at the P < 0.05 level.
* One Way ANOVA Test, **Post-hoc Bonferroni Test.
Bold P-values indicate statistically signifi cant.
Materials and methods
Study design
This retrospective case-control study was conducted based on the electronic records of pregnant women diagnosed with preeclampsia and HELLP syndrome between January 2017 and December 2022 in our hospital, which is a tertiary care center for high-risk pregnancies. The Mersin University Clinical Research Ethics Committee approved the study (Decision No. 2023/39) and informed consent was obtained from all participants.
Patient selection
Two separate study groups were dia g nosed with preeclampsia or HELLP syndrome, and pregnant women without these diagnoses were considered the control group. The control group consisted of healthy pregnant women whose gestational ages were matched, without obstetric pathology, any maternal medical complication, or autoimmune disorder history during the same period. Exclusion criteria are chronic additional diseases such as diabetes mellitus, chronic hypertension, thromboembolism, thrombophilia, hepatic or renal disease history, multiple pregnancy, membrane rupture, presence of any infection, pregnancies with assisted reproductive techniques, and presence of a fetal anomaly.
Basic data collected for analysis from electronic medical records include age and obstetric history, hemoglobin, hematocrit, leukocytes, neutrophils, lymphocytes, monocytes, and platelet counts; mean platelet volume (MPV); plateletcrit (PCT); alanine aminotransferase (ALT); aspartate aminotransferase (AST); lactate dehydrogenase (LDH); uric acid levels; DNI; SII; platelet lymphocyte ratio (PLR); monocyte lymphocyte ratio (MLR); neutrophil lymphocyte ratio (NLR); proteinuria results; week of birth; birth weights of newborns; 1st and 5th min Apgar scores; and gestational weeks were calculated according to the last menstrual period or, for those who did not know the last menstrual period, according to fetal ultrasonography measurements in the first trimester. The data at the diagnosed gestational week were recorded. The dia g nosis of preeclampsia and HELLP syndrome was accepted according to the criteria of the American College of Obstetricians and Gynecologists (ACOG), which was reported in 2020 [12]. For the diagnosis of preeclampsia, systolic blood pressure ≥ 140 mmHg and/or diastolic blood pressure ≥ 90 mmHg in two measurements 4 h apart after the 20th gestational week in a woman without pre-existing hypertension and various multisystem disorders (platelet count less than 100,000 × 109/L with or without new-onset proteinuria; elevation of liver enzymes up to twice the upper limit of normal concentration; unexplained severe persistent right upper quadrant or epigastric pain; serum creatinine ≥ 1.1 mg/dL or doubling of serum creatinine concentration in the absence of kidney disease; pulmonary edema; or unexplained new-onset headache unresponsive to acetaminophen) were used [12]. For the diagnosis of HELLP syndrome, increased LDH values, AST and ALT values increased more than twice the upper limit of the normal range, and platelet count was less than 100,000 × 109/L [12]. Hemogram, bio chemistry, and full urinalysis tests are requested and recorded for all pregnant women who apply to our hospital for delivery. These data for the study and control groups were obtained from hospital records. The serum DNI value is obtained using an automated cell analyzer and is routinely calculated within the hemogram parameters in our hospital. This system is a flow cytometry-based hematologic analyzer that uses two independent WBC counting methods: a myeloperoxidase (MPO) channel and a lobularity/nuclear density channel. DNI is an inflammatory marker calculated by a flow cytometry-based hematological analyzer using the following formula: DNI (%) = (leukocyte subfraction assayed in the MPO channel by the cytochemical reaction) + (leukocyte subfraction assayed using the nuclear lobularity channel by reflected light beam measurements) [11]. SII was calculated and recorded with the formula (neutrophil × platelet × lymphocyte). Complete blood count parameters were determined using ADVIA 2120i (Siemens Healthcare), and bio chemical parameters were determined using Beckman Coulter AU680 and AU480 devices.
Statistical analysis
Statistical Package for the Social Sciences (Version 22, SPSS Inc., Chicago, IL, USA) was used to analyze the data. Kolmogorov-Smirnov Test, Shapiro-Wilk Test and histograms were used to evaluate the normality of distributions. In the analysis of the data, the mean and standard deviation, median, minimum, and maximum values of the features were used while performing continuous data statistics. One Way ANOVA and Kruskal Wallis Test statistics were used to compare the means of three independent groups. If there was a difference between the means in group measurements, pairwise comparisons were evaluated with the Bonferroni corrected Mann-Whitney U-Test and Post-hoc Bonferroni Test. The relationship of inflammatory parameters with each other was evaluated with the Pearson‘s correlation. Receiver operating characteristic (ROC) analysis was performed to evaluate the predictive value of the DNI for the development of preeclampsia and HELLP syndrome. The statistical significance level of the data was taken as P < 0.05.
Results
121 pregnant women who met the inclusion and exclusion criteria were included in the study. In the comparison of sociodemographic, laboratory, and neonatal characteristics of the groups (Tab. 1), maternal age, hematocrit, lymphocytes, and MPV were similar between groups (P > 0.05). There was a statistically significant difference between the HELLP group and the control group in terms of gravida, parity, and 1st minute Apgar score (P < 0.05), but there was no significant difference between the other groups (P > 0.05). There was a statistically significant difference between the HELLP group and the other two groups in terms of platelet, leukocyte, neutrophil, PCT, AST, ALT values, and 5th minute Apgar score (P < 0.05); there was no significant difference between the control and preeclampsia groups (P > 0.05). There was a statistically significant difference between all groups in terms of LDH value, gestational week, and birth weight (P < 0.05). The difference between the control group and the preeclampsia and HELLP groups in terms of proteinuria and uric acid values was statistically significant (P < 0.05), but no significant difference was found between the preeclampsia and HELLP groups (P > 0.05). There was a statistically significant difference between the preeclampsia and control groups in terms of hemoglobin and monocyte values (P < 0.05), but there was no significant difference between the other groups (P > 0.05).
In the comparison of the systemic inflammatory parameters of the groups (Tab. 2), NLR was higher in the HELLP group, and there was a statistically significant difference when compared with other groups (P < 0.05). NLR was similar in the control and preeclampsia groups (P > 0.05). It was observed that MLR was lower in the preeclampsia group, and there was a statistically significant difference when compared with other groups (P < 0.05). MLR was similar in the control and HELLP groups (P > 0.05). It was observed that PLR was lower in the HELLP group, and there was a statistically significant difference when compared with other groups (P < 0.05). PLR was similar in the control and preeclampsia groups (P > 0.05). It was observed that DNI was higher in the HELLP group, and there was a statistically significant difference when compared with other groups (P < 0.05). It was determined that the preeclampsia group had a higher DNI value compared to the control group, but the difference was not significant (P > 0.05). All groups were similar in terms of SII (P > 0.05).
In the correlation of inflammatory parameters (Tab. 3), NLR and PLR were moderately positively correlated (R = 0.444; P < 0.001). MLR has a weak positive correlation with NLR (R = 0.439; P < 0.001), but not significantly with PLR. DNI has a moderate positive correlation with NLR (R = 0.535; P < 0.001) and a weak positive correlation with SII (R = 0.281; P = 0.002). SII has a strong positive correlation with NLR (R = 0.781; P < 0.001) and PLR (R = 0.778; P < 0.001) and a moderately positive correlation with DNI (R = 0.281; P = 0.002).
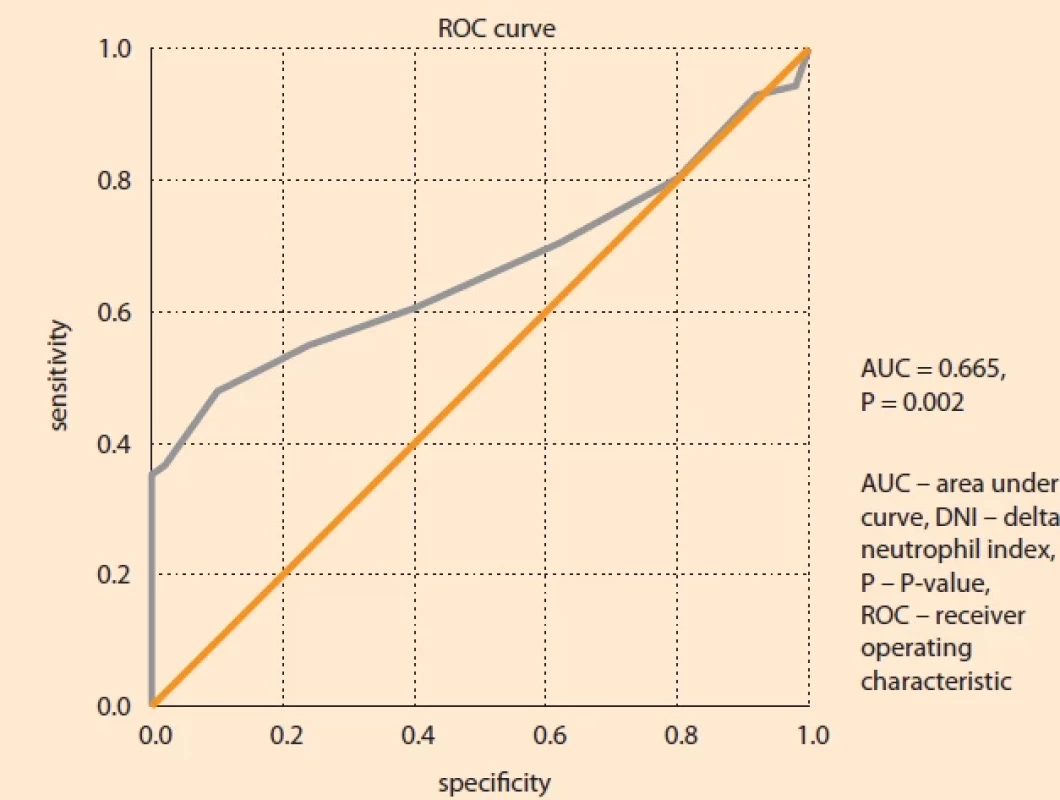
In determining the cut-off for DNI in preeclampsia and HELLP syndrome (Fig. 1, Tab. 4), the optimal cut-off value is P > 0.8 and the AUC value is 0.665 (95% CI 0.569–0.760; P = 0.002), showing moderate accuracy. At this cut-off value, the sensitivity of the test is 47.89%, the specificity is 90.00%, the positive predictive value is 87.2%, and the negative predictive value is 54.9%.
Discussion
Although some factors responsible for the etiology of preeclampsia and HELLP syndrome are known, the etiopathogenesis has not been fully clarified. New studies on this situation continue to be carried out. Inflammation is considered to be responsible for the emergence of many diseases. Preeclampsia and HELLP syndrome are also thought to be systemic syndromes characterized by inflammatory and anti-angiogenic conditions [13]. In this study, serum levels of serum DNI and SII parameters were investigated in preeclampsia and HELLP syndrome. In the studies available in the literature, these inflammatory parameters were investigated only in preeclampsia. This study is the first to investigate serum DNI and SII parameters in HELLP syndrome with preeclampsia. In our study, we concluded that inflammatory markers are significantly different in HELLP syndrome, inflammation may play a role in the pathogenesis of these diseases, and serum DNI values can be used in the diagnosis.
Placenta and trophoblast cells play an important role in the development of preeclampsia. Abnormal placental development and trophoblast invasion during placental development reduce placental perfusion [14]. Abnormal trophoblast invasion reduces arterial blood flow, resulting in oxidative damage and inflammation as a result of the hypoxic environment [15]. Excessive inflammation and angiogenic imbalance are thought to be the causes of preeclampsia [13].
Pregnancy is a successful semi-allograft reaction. The balance between immune and inflammatory responses plays an important role in maintaining pregnancy. Ensuring this balance; it provides blastocyst implantation, trophoblast cell proliferation and differentiation, and placental development [16]. In preeclampsia, this favorable environment is disrupted, and an overactive inflammatory response occurs [17]. In a study, it was concluded that the development of preeclampsia can be reduced by suppressing the inflammatory response [18].
Endothelial dysfunction is thought to be the main cause of the pathogenesis of preeclampsia. Oxidative stress triggered in the placenta as a result of impaired trophoblast invasion increases the synthesis of proinflammatory precursors and increases the inflammatory response. Increased inflammatory responses cause vascular endothelial dysfunction [19,20]. Endothelial dysfunction is accompanied by high levels of inflammatory parameters [21]. As a result of immune dysfunction, chronic inflammation occurs. Production of reactive oxygen species is induced, and endothelin-1 expression is increased. As a result of an increased inflammatory response, inflammatory cells accumulate in vascular areas, and vascular functions are impaired [22].
Although the pathophysiological mechanisms of preeclampsia and HELLP syndrome remain unclear, this suggests that inflammatory events may play a role in the formation mechanism of these diseases by causing placental changes. In recent years, important studies have been carried out on this subject. In a study evaluating the systemic inflammatory response in preeclampsia, inflammatory markers such as tumor necrosis factor-alpha, interleukin-6, and C-reactive protein were evaluated. They found that these markers changed significantly in the preeclampsia group compared to controls [23]. Yiyenoglu et al. found similar results in their study. They found that oxidative stress resulting from placental reperfusion increased the levels of tumor necrosis factor-alpha, interleukin-6, interleukin-10, and C-reactive protein, and this led to an intravascular inflammatory response and endothelial dysfunction [24].
NLR, PLR, MLR, and SII are simple and inexpensive markers of systemic inflammation that are frequently used to assess the inflammatory response. These parameters were investigated in some studies to evaluate the inflammatory response in patients with preeclampsia. Bektas et al. found that NLR, PLR, and MLR values in preeclampsia and severe preeclampsia showed significant differences compared to healthy controls. However, these values were not found to be statistically significant between the preeclampsia and severe preeclampsia groups [25]. In the study of Wang et al., it was observed that the preeclampsia group had higher NLR and MLR values compared to the control group. These values were found to be significantly higher in the severe preeclampsia group [26]. In a study involving 2,050 pregnant women, they found that NLR and PLR values were similar before the 20th gestational week, and high NLR and lower PLR values were found in preeclamptic patients just before delivery [27]. Gezer et al. found that NLR and PLR values were high in patients with preeclampsia and stated that NLR was the strongest predictive variable [28]. However, in another study, it was reported that NLR and PLR values failed to predict the progression of preeclampsia [29]. In our study, we found that the NLR value was higher and the PLR value was lower, especially in HELLP syndrome. We concluded that there was no change in these parameters in preeclampsia patients. We observed that the MLR value was significantly lower in patients with preeclampsia than in other groups, and it was not different in HELLP syndrome compared to healthy controls. We found that SII did not change in all groups.
DNI is the immature granulocyte fraction calculated by a blood cell analyzer [30]. Since it is easily accessible, inexpensive, reproducible, fast, and accurate to measure, it has been a parameter used for both the presence and severity of inflammation in recent years [30]. In recent years, some studies have been conducted on DNI as an inflammatory bio marker in obstetrics [7,8,10,11]. Serum DNI values have also been studied in preeclamptic patients. In one study, it was found that there was a significant difference in DNI between the normal pregnancy group and the preeclampsia group. The DNI was observed to be significantly higher in the severe preeclampsia group compared to the healthy pregnancy group and the mild preeclampsia group. It has been found that it is an adequate marker to determine the severity of preeclampsia in preeclamptic patients [31]. In the same study, the relationship between DNI and clinical signs of preeclampsia was evaluated, and it was shown that there was a positive correlation between DNI and symptoms [31]. In the study of Ozkan et al., it was concluded that there was no significant difference in terms of DNI in the control, gestational hypertension, preeclampsia, and severe preeclampsia groups [32]. In our study, we concluded that the DNI value was higher in patients with preeclampsia compared to the control group, but the difference was not significant. This value was higher in patients with HELLP syndrome, and the difference was significant between the groups. We found that when the DNI value is above 0.8, it can predict preeclampsia and HELLP syndrome.
Limitations of our study: retrospective design, single center, and small sample size. Conducting the study in a tertiary center and selecting higher-risk pregnancies to be included in the study constitutes the strength of the study. The fact that the analysis of the received data was from a single central laboratory prevented variability. The most important strength of the study was that the investigated hematological and bio chemical indices were used for the first time in HELLP syndrome patients, besides preeclampsia.

*Correlation is signifi cant at the 0.01 level (2-tailed), **Correlation is signifi cant at the 0.05 level (2-tailed).
Bold P-values indicate statistically signifi cant.

Conclusion
We think that inflammation may be involved in the pathogenesis of preeclampsia and HELLP syndrome, as well as in the pathogenesis of many diseases, and that maternal serum DNI values can help the diagnosis when used together with other bio chemical parameters. However, large-scale prospective studies including other possible risk factors may be useful to confirm these results.
Authorship
YD, ŞK: concept, author, design, data processing, analysis, and interpretation
YD, FA: analysis and interpretation, design, statistical analysis
ŞK, KA: data collection, processing, analysis
AC, YD: audit, statistical analysis, design
AC, FA: control, concept
All authors approved the final version of the manuscript.
ORCID authors
Y. Dal 0000-0001-7162-4644
Ş. Karagün 0000-0003-2339-1609
F. Akkuş 0000-0001-7037-9165
K. Akay 0000-0002-6098-2259
A. Coşkun 0000-0002-2887-9649
Submitted/Doručeno: 3. 7. 2023
Accepted/Přijato: 10. 8. 2023
Yusuf Dal, MD
Department of Obstetrics and Gynecology
Perinatology Department
Mersin University Faculty of Medicine
TR-33110 Mersin, Turkey
dryusufdal@gmail.com
Sources
1. Metz TD, Berry RS, Fretts RC et al. Obstetric care consensus #10: management of stillbirth: (replaces practice bulletin number 102, March 2009). Am J Obstet Gynecol 2020; 222 (3): B2–b20. doi: 10.1016/j.ajog.2020.01.017.
2. López-Jaramillo P, García RG, López M. Preventing pregnancy-induced hypertension: are there regional differences for this global problem? J Hypertens 2005; 23 (6): 1121–1129. doi: 10.1097/01.hjh.0000170371.49010.4a.
3. Szarka A, Rigó J Jr, Lázár L et al. Circulating cytokines, chemokines and adhesion molecules in normal pregnancy and preeclampsia determined by multiplex suspension array. BMC Immunol 2010; 11 : 59. doi: 10.1186/1471-21 72-11-59.
4. Roberts JM, Hubel CA. The two stage model of preeclampsia: variations on the theme. Placenta 2009; 30 (Suppl A): S32–S37. doi: 10.1016/j.placenta.2008.11.009.
5. Lyall F, Greer IA, Boswell F et al. The cell adhesion molecule, VCAM-1, is selectively elevated in serum in pre-eclampsia: does this indicate the mechanism of leucocyte activation? Br J Obstet Gynaecol 1994; 101 (6): 485–487. doi: 10.1111/j.1471-0528.1994.tb13 146.x.
6. Ahn C, Kim W, Lim TH et al. The delta neutrophil index (DNI) as a prognostic marker for mortality in adults with sepsis: a systematic review and meta-analysis. Sci Rep 2018; 8 (1): 6621. doi: 10.1038/s41598-018-24211-7.
7. Şahin Uysal N, Eroğlu H, Özcan Ç et al. Is the serum delta neutrophil index level different in gestational diabetic women? J Matern Fetal Neonatal Med 2020; 33 (19): 3349–3354. doi: 10.1080/14767058.2020.1760833.
8. Cho HY, Jung I, Kwon JY et al. The Delta Neutrophil Index as a predictive marker of histological chorioamnionitis in patients with preterm premature rupture of membranes: a retrospective study. PLoS One 2017; 12 (3): e0173382. doi: 10.1371/journal.pone.0173382.
9. Doğru Ş, Akkuş F, Atci AA et al. The role of the systemic inflammatory index in determining the length of hospital stay in patients with hyperemesis gravidarum. Ceska Gynekol 2023; 88 (3): 172–178. doi: 10.48095/cccg2023 172.
10. Doğru Ş, Atci AA, Akkuş F et al. Predictability of hematological parameters in the diagnosis of cesarean scar pregnancy. J Lab Physicians 2023; 15 (3): 425–430. doi: 10.1055/s-0043-1761 929.
11. Eroğlu H, Şahin Uysal N, Sarsmaz K et al. Increased serum delta neutrophil index levels are associated with intrahepatic cholestasis of pregnancy. J Obstet Gynaecol Res 2021; 47 (12): 4189–4195. doi: 10.1111/jog.15028.
12. Gestational hypertension and preeclampsia: ACOG practice bulletin, number 222. Obstet Gynecol 2020; 135 (6): e237–e260. doi: 10.1097/AOG.0000000000003891.
13. de Oliveira LG, Karumanchi A, Sass N. Preeclampsia: oxidative stress, inflammation and endothelial dysfunction. Rev Bras Ginecol Obstet 2010; 32 (12): 609–616. doi: 10.1590/ s0100-72032010001200008.
14. Karthikeyan VJ, Lip GY. Endothelial damage/dysfunction and hypertension in pregnancy. Front Biosci (Elite Ed) 2011; 3 (3): 1100–1108. doi: 10.2741/314.
15. Myatt L, Webster RP. Vascular bio logy of preeclampsia. J Thromb Haemost 2009; 7 (3): 375–384. doi: 10.1111/j.1538-7836.2008.03259.x.
16. Mor G, Kwon JY. Trophoblast-microbio me interaction: a new paradigm on immune regulation. Am J Obstet Gynecol 2015; 213 (4 Suppl): S131–S137. doi: 10.1016/j.ajog.2015.06.039.
17. Harmon AC, Cornelius DC, Amaral LM et al. The role of inflammation in the pathology of preeclampsia. Clin Sci (Lond) 2016; 130 (6): 409–419. doi: 10.1042/CS20150702.
18. Subha M, Pal P, Pal GK et al. Decreased baroreflex sensitivity is linked to sympathovagal imbalance, low-grade inflammation, and oxidative stress in pregnancy-induced hypertension. Clin Exp Hypertens 2016; 38 (8): 666–672. doi: 10.1080/10641963.2016.1200596.
19. Tenório MB, Ferreira RC, Moura FA et al. Cross-talk between oxidative stress and inflammation in preeclampsia. Oxid Med Cell Longev 2019; 2019 : 8238727. doi: 10.1155/2019/8238 727.
20. Michalczyk M, Celewicz A, Celewicz M et al. The role of inflammation in the pathogenesis of preeclampsia. Mediators Inflamm 2020; 2020 : 3864941. doi: 10.1155/2020/3864941.
21. Wang Y, Gu Y, Zhang Y et al. Evidence of endothelial dysfunction in preeclampsia: decreased endothelial nitric oxide synthase expression is associated with increased cell permeability in endothelial cells from preeclampsia. Am J Obstet Gynecol 2004; 190 (3): 817–824. doi: 10.1016/j.ajog.2003.09.049.
22. Granger JP, Alexander BT, Llinas MT et al. Pathophysiology of hypertension during preeclampsia linking placental ischemia with endothelial dysfunction. Hypertension 2001; 38 (3 Pt 2): 718–722. doi: 10.1161/01.hyp.38.3.718.
23. Mihu D, Razvan C, Malutan A et al. Evaluation of maternal systemic inflammatory response in preeclampsia. Taiwan J Obstet Gynecol 2015; 54 (2): 160–166. doi: 10.1016/j.tjog.2014.03. 006.
24. Yiyenoğlu ÖB, Uğur MG, Özcan HÇ et al. Assessment of oxidative stress markers in recurrent pregnancy loss: a prospective study. Arch Gynecol Obstet 2014; 289 (6): 1337–1340. doi: 10.1007/s00404-013-3113-4.
25. Bektaş O, Bektaş K, Taşın C. The role of systemic inflammatory indexes in predicting preeclampsia and its severity. Perinatal J 2019; 27 (2): 62–67. doi: 10.2399/prn.19.0272003.
26. Wang J, Zhu QW, Cheng XY et al. Assessment efficacy of neutrophil-lymphocyte ratio and monocyte-lymphocyte ratio in preeclampsia. J Reprod Immunol 2019; 132 : 29–34. doi: 10.1016/j.jri.2019.02.001.
27. Mannaerts D, Heyvaert S, De Cordt C et al. Are neutrophil/lymphocyte ratio (NLR), platelet/lymphocyte ratio (PLR), and/or mean platelet volume (MPV) clinically useful as predictive parameters for preeclampsia? J Matern Fetal Neonatal Med 2019; 32 (9): 1412–1419. doi: 10.1080/14767058.2017.14710701.
28. Gezer C, Ekin A, Ertas IE et al. High first-trimester neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios are indicators for early dia g nosis of preeclampsia. Ginekol Pol 2016; 87 (6): 431–435. doi: 10.5603/GP.2016.0021.
29. Yavuzcan A, Cağlar M, Ustün Y et al. Mean platelet volume, neutrophil-lymphocyte ratio and platelet-lymphocyte ratio in severe preeclampsia. Ginekol Pol 2014; 85 (3): 197–203.
30. Nahm CH, Choi JW, Lee J. Delta neutrophil index in automated immature granulocyte counts for assessing disease severity of patients with sepsis. Ann Clin Lab Sci 2008; 38 (3): 241–246.
31. Cho HY, Jung I, Kim SJ et al. Increased delta neutrophil index in women with severe preeclampsia. Am J Reprod Immunol 2017; 78 (3). doi: 10.1111/aji.12705.
32. Ozkan D, Ibanoglu MC, Adar K et al. Efficacy of blood parameters in predicting the severity of gestational hypertension and preeclampsia. J Obstet Gynaecol 2023; 43 (1): 2144175. doi: 10.1080/01443615.2022.2144175.
Labels
Paediatric gynaecology Gynaecology and obstetrics Reproduction medicineArticle was published in
Czech Gynaecology

2023 Issue 6
-
All articles in this issue
- Is there a difference between acute appendicitis in pregnant and non-pregnant women?
- Are the serum delta neutrophil index and systemic inflammatory index useful as predictive parameters for preeclampsia and HELLP syndrome?
- Medical Termination of Pregnancy (MToP) in the 1st trimester – the role of human chorionic gonadotropin and ultrasound in pregnancy diagnosis and MToP follow-up
- Pregnancy termination indications and outcomes before 24 weeks of gestation – a case series
- An updated perspective on diagnostics and treatment of idiopathic granulomatous mastitis
- A rare case of a live abdominal pregnancy in a woman with subsequent gestation in utero
- DNA analysis of partial hydatidiform mole revealing triandric monogynic tetraploidy
- Outcome of a patient with Herlyn-Werner-Wunderlich syndrome treated with Balloon septostomy – pre- and postsurgical ultrasound findings
- Methods and techniques of fertility preservation in patients with endometriosis
- Assisted oocyte activation
- Effect of pelvic floor status on the outcome of pelvic organ prolapse surgery
- Preeclampsia and diabetes mellitus
- Diagnosis of complicated gynaecological inflammations by computed tomography – one center experience
- Emergent hysterectomy for placenta accreta spectrum – some additional techniques
- Diagnostika a léčba endometriózy: Doporučený postup Sekce pro léčbu endometriózy ČGPS ČLS JEP
- Czech Gynaecology
- Journal archive
- Current issue
- About the journal
Most read in this issue
- Diagnostika a léčba endometriózy: Doporučený postup Sekce pro léčbu endometriózy ČGPS ČLS JEP
- Preeclampsia and diabetes mellitus
- Assisted oocyte activation
- Is there a difference between acute appendicitis in pregnant and non-pregnant women?
